Enhanced Medical Expertise and
Patient Perspective
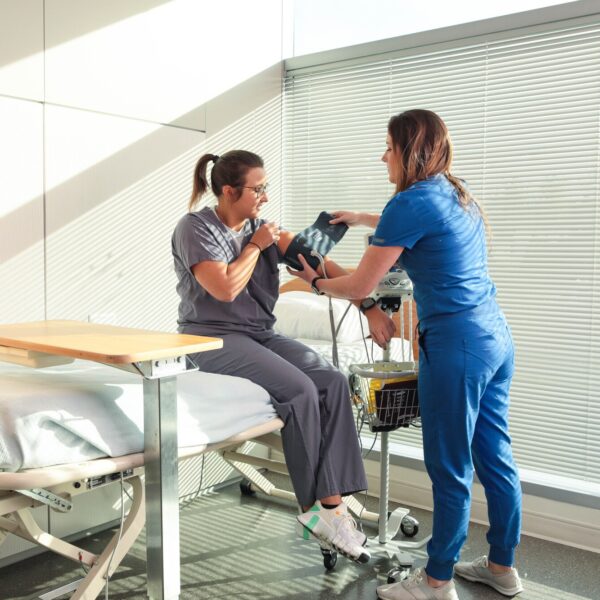
Our medical experts are supported by a team of advanced clinical practitioners (ACPs) whose unique perspective brings added value to the clinical development team. These healthcare professionals are highly trained experts with years of hands-on experience in both patient care and clinical research. They are not only registered nurses with advanced academic education, but they also have additional training as medical providers and must be licensed as advanced practice registered nurses. The years of hands-on experience our ACPs bring in patient care ensure that the patient perspective is acknowledged throughout the trial.
Fully Integrated into Your Study
Our team of global clinical practitioners are fully integrated into your study – supporting our medical and operations team members while providing that additional layer of knowledge and insight that will streamline your study – addressing potential challenges early on in the study planning. Utilizing their clinical backgrounds, they can:
- Expand and enrich in-house medical expertise
- Provide an additional layer of practical knowledge and clinical expertise, and insights that can proactively address potential protocol challenges
- Bring patient-centric and site-centric perspectives to your study – ensuring both the protocol is feasible from a site standpoint and patient experience is enhanced
- Leverage clinical logistical knowledge and expertise to support all aspects of study conduct

- Ongoing team training and education
- Identifies potential risks with protocol implementation
- Provides patient-centric protocol considerations

- Recruitment strategy insight and advice
- Assists with operational questions
- Identifies potential clinical challenges and recommends solutions

- Reviews CRF for appropriate language and ease of use
- Serves as a resource to the data management team for clinical related questions

- Assists with safety narratives and analysis of safety events
- Assists with review of safety and laboratory data
Learn More from Our Team of ACPs
Our team of highly trained experts have years of hands on experience in both patient care and clinical research across a wide range of therapeutic areas. Watch the videos below to learn more.