Keeping Trials on Pace by Effectively
Identifying, Mitigating and Monitoring Risk
The Medpace monitoring approach is a holistic, adaptive process, evaluated on an ongoing basis for the life of the trial. It directs focus and monitoring/oversight activities to the areas of greatest risk.
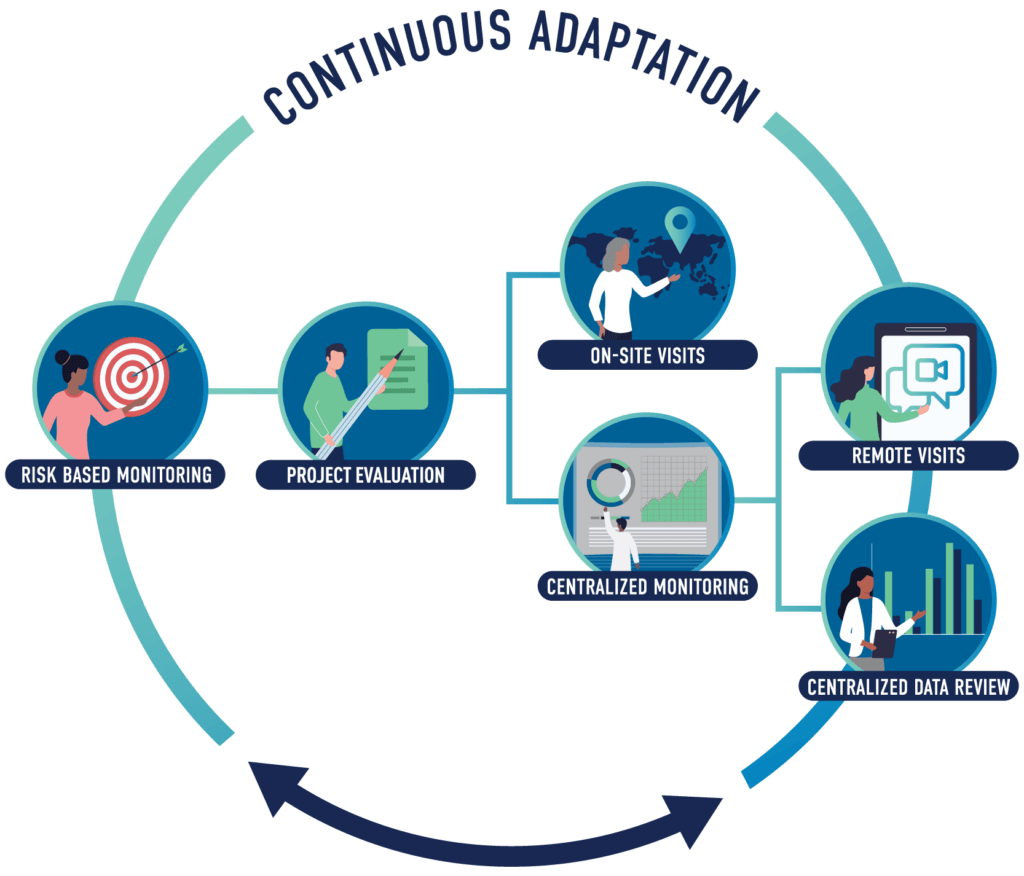
Risk assessment begins with a thorough protocol evaluation that drives specific activities including centralized data reviews and remoting monitoring visits, as well as on-site monitoring visits.
Adaptive Monitoring Capabilities
Medpace identifies protocol elements that have the greatest potential to impact subject safety and data integrity and adapts the monitoring approach accordingly. The key components include:
- Protocol Evaluation: Focuses on identifying critical study aspects, risks, and potential mitigation strategies
- Centralized Monitoring: Ensures early identification of quality, safety, and operational risks via centralized data reviews and remote monitoring visits
- On-site Visits: Allows for source review unable to be achieved remotely and in-person site support and education/re-education to address needs identified via centralized review
One of the Medpace advantages is the level of clinical review given to protocol risk assessment. In addition to the operations team, our Medical Monitors, Advanced Nurse Practitioners, and Clinical Data Reviewers provide scientific insights and practical perspectives that result in a more comprehensive, effective monitoring strategy.
The Medpace Advantage in Clinical Monitoring
Risked Based Monitoring (RBM) Framework
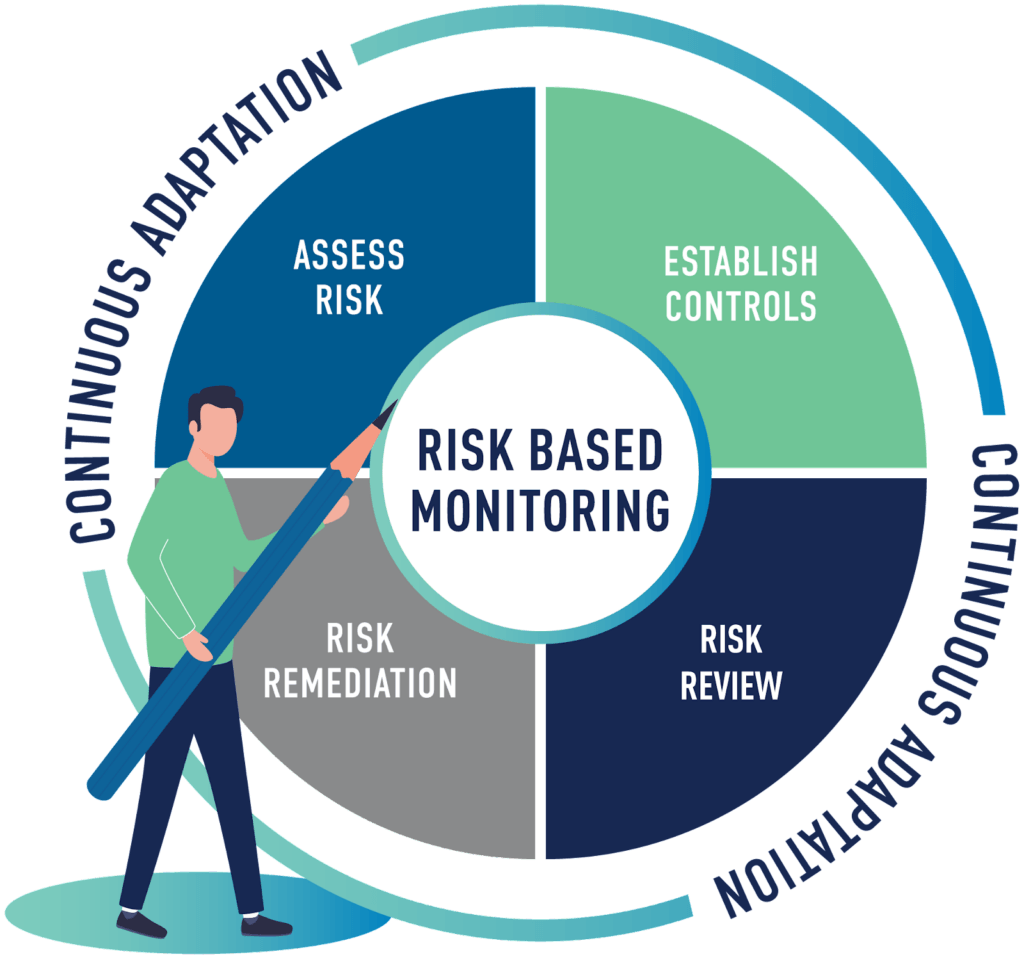
We apply our RBM framework to each study to deliver the most efficient monitoring strategy while protecting the welfare of patients and data integrity. The framework has four primary stages enhanced by continuous adaptation:
- Protocol Evaluation: A thorough protocol review will help determine the right combination of remote, on-site, and centralized monitoring.
- Establish Controls: Controls are inherent in Medpace SOPs, which are then further customized and detailed throughout the Project Governing Documentation, Project Plans and Project Training.
- Centralized Monitoring: Ongoing oversight activities, including centralized data reviews for both medical surveillance and operational oversight.
Risk Remediation: Risk identified by centralized monitoring results in immediate action such as additional monitoring activities, site training, and/or study plan modifications, taken to mitigate risk, maintain compliance and ensure the continued success of the study.
Technology: Real Time Metrics
& Actionable Outputs
ClinTrak® Data Explorer provides an interactive analytics platform that combines multiple sources of unstructured data into easy to use dashboards. Data sources are integrated so that large amounts of information can be reviewed in aggregate. Examples of data sources include central lab, EDC, IRT, Electronic Health Records, eConsent, eDiaries, and direct-to-patient services including home health, study product and/or supplies, and travel support. Through our platform, we can shape and visualize the data to identify trends, outliers and other points of interest. This allows our teams to quickly visualize and assess a large volume of data—whether it be operational, clinical, or safety data—to support forward-looking study decisions. Additionally, the self-service interactive dashboards allow study teams to easily customize and share their findings. Integrated data allows for the development of:
- Customized study-specific dashboards based on unique project risks
- Reports and visualizations at patient, site, country, study, and program levels to perform review of pertinent data and allow for analysis of trends and outliers
ClinTrak Data Explorer allows our team to identify anomalies, mitigate risk, and escalate issues as necessary, and provides on-demand access for internal and external user oversight. For Clinical Research Associates (CRAs), this enables more monitoring to occur remotely, reducing the frequency of on-site visits while still maintaining monitoring effectiveness. CRAs can focus on site education/support and quality driven by technology/centralized data review.
Cross-Functional Collaboration
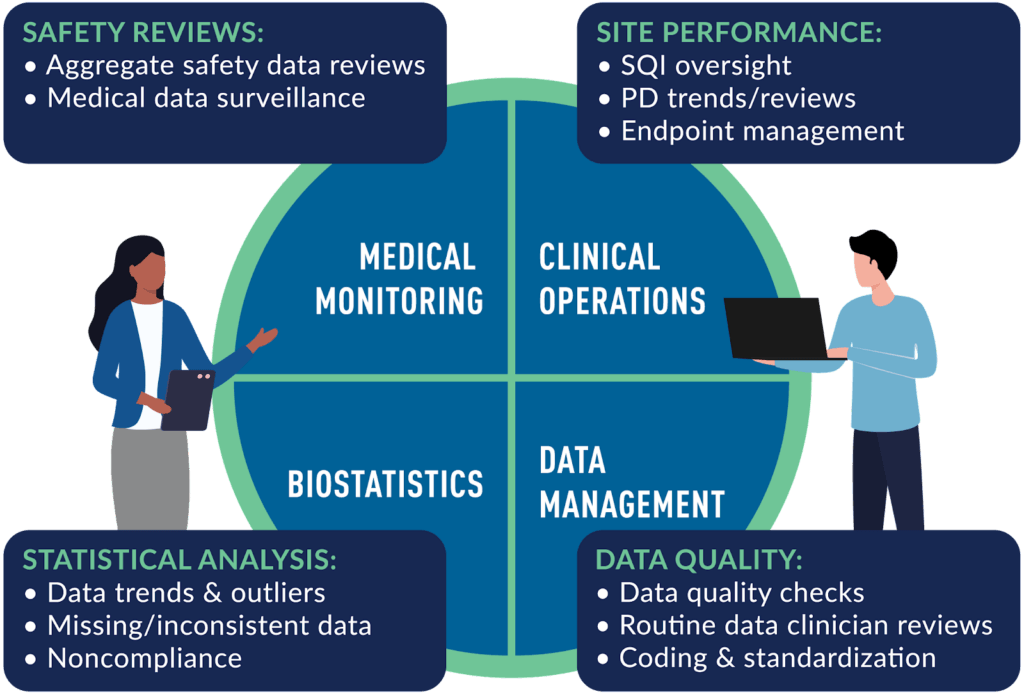
Risks can arise at any stage of a study. We provide cross-functional expertise to identify, mitigate and monitor risk throughout the study duration. A key differentiator of Medpace is the extensive involvement of our physicians throughout study conduct. RBM is no exception as medical surveillance and ongoing medical reviews are led by Medical Monitors and supported by Advanced Nurse Practitioners, safety coordinators and a dedicated team of data clinicians who follow each patient through his/her study experience. The study leadership team of Medical Monitor and Clinical Trial Manager ensures a continued focus on clinical as well as operational data.
Sites & Patient Focus
One approach does not fit all; we partner with sites to develop custom monitoring strategies that best align with their capabilities.
With over 30 years of experience, Medpace has developed and maintained strong working relationships with thousands of investigators and research sites. Avoiding site burden is critical in fostering collegial relationships and ensuring sound study execution. As we implement study-level monitoring changes, we work closely with sites to adapt strategies that best align with their specific processes.
Reducing patient burden is also critical in designing studies. The shift to decentralized services has had a tremendous impact on patient engagement and the management of study requirements. Medpace has developed tools and processes to further these initiatives to minimize the inconvenience of study participants while enhancing protocol compliance.
CRA Training & Development
– A Hallmark of Success
Well-trained CRAs play a crucial part in the success of clinical trials, which is why we invest heavily in a comprehensive and rigorous internal training program called PACE® (Professionals Achieving CRA Excellence). CRA education does not end with the initial training of our associates but continues throughout their tenure at Medpace.



