Specialized Expertise to Accelerate Complex Ophthalmology Clinical Trials
Ophthalmology clinical trials are becoming more complex due to advanced imaging requirements, rare disease recruitment challenges, evolving endpoints, and increased development of gene and cell therapies. These trials require a clinical research organization (CRO) partner with innovative patient enrollment capabilities, strong relationships with top-performing sites, strategic study design and execution, and RNA based therapy experience.
Accelerate your path to approval with guidance from our global team of ophthalmology-focused medical, operational, and regulatory experts. Leveraging our extensive experience, world-class expertise, and consistent track record of success, we offer the flexibility to adapt to the unique needs of each ophthalmology trial, overcome complex challenges, and keep your trial on time and within budget.
Making the Complex Seamless® in Global Ophthalmology Clinical Trials
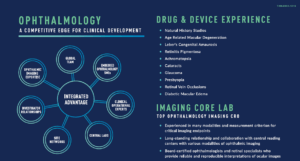
Drive the successful execution of Phase I-IV ophthalmology clinical trials through a partner with strong communication, coupled with early planning and experience collaborating with global regulatory authorities. Sponsors from emerging biotechs to global pharmaceutical companies have trusted Medpace for over 30 years to lead their ophthalmology development across a wide range of indications, including but not limited to Age-Related Macular Degeneration, Inherited Retinal Disease, Cataracts, Glaucoma, Cornea, Diabetic Macular Edema, and Natural History Studies.

Leading Ophthalmology CRO
Our medical, operations, and regulatory staff understand the complexities of ophthalmology trials from the perspective of the Sponsor, the clinical investigator, and the reviewer at the regulatory agencies; we bring these perspectives to each clinical trial that we conduct to help keep trials running efficiently and according to protocol.
By partnering with a global CRO with a cross-functional collaborative model, the Medpace ophthalmology team can support your clinical trials through:
- Providing comprehensive support via our dedicated global regulatory submissions and global regulatory affairs teams
- Extensive experience partnering with the Sponsor’s choice of vendor for ophthalmology imaging to support each trial’s unique needs
- CAP-accredited central laboratory services for safety testing, PK/PD, biomarkers, and genomic testing; ensure the perfect harmonization of global data through wholly-owned and purpose-built laboratories with state-of-the-art infrastructure
Our Proven Site Start-Up Model
In this two-minute overview, discover how Medpace’s Ophthalmology team accelerates trials through a customized start-up approach designed to activate sites faster and shorten study timelines.
Strong, Long-Term Relationships in Ophthalmic Indications
Medpace has strategically built strong, long-term relationships with investigators, top-performing sites, networks, and key opinion leaders around the world that specialize in ophthalmic indications. The long-standing relationships combined with our experience help facilitate strategic site selection, start-up, and recruitment efforts translating into the successful and timely recruitment, conduct, and delivery of a study.
Gene Therapy Research in Ophthalmology Clinical Trials
Executing ophthalmology gene therapy clinical trials is complex due to challenges including study procedures and assessments, site training, and sample storage. Medpace’s expertise and lessons learned in gene therapy ophthalmology clinical trials allow us to provide guidance to help Sponsors successfully navigate common complexities. This will ensure successful patient recruitment and retention, effective investigator training, and reduce trial risks through our in-depth experience in gene therapy research across multiple therapeutic areas with both subretinal and intravitreal delivery.
Proactive Patient Recruitment & Retention in Ophthalmology Clinical Trials
The success of clinical trials is wholly dependent on the success of sites and the engagement of patients. Our proprietary IntelliPACE® feasibility strategy is informed by a multitude of internal and external sources to provide a highly customized, informed recruitment strategy for each study. Accelerate patient enrollment with strategic guidance from our dedicated, in-house patient recruitment and retention teams who are experienced in recruiting complex patient populations including pediatrics, geriatrics, and rare ocular diseases.

Key Ophthalmology Disease Indications
Our global team of experienced medical, clinical, and regulatory experts have been involved in the management and execution of ophthalmology trials around the world.
In-house ophthalmologists with deep backgrounds in conducting complex studies are embedded into your program from project initiation to completion, providing hands-on leadership and translating medical knowledge into execution.
Medpace has experience across a broad range of indications, including but not limited to:
- Inherited retinal diseases
- Diabetic retinopathy
- Wet AMD
- Geographic atrophy
- Orbital Disease (TED)
- Retinal gene therapy
- Ocular Surface Disease
- Glaucoma
Article
TRENDS AND
BREAKTHROUGHS IN THE WET & DRY
AMD CLINICAL TRIAL LANDSCAPE
Global Ophthalmology Clinical Trial Experts
Accelerate your path to approval by partnering with Medpace’s global, cross-functional teams, including ophthalmology-specialized clinical trial managers and project coordinators that provide knowledgeable training to sites and operational leadership.
Medpace’s in-house, board-certified ophthalmologists and gene therapy specialists with 76+ years of combined experience add an additional layer of medical and scientific expertise and are embedded within the project team throughout the lifecycle of your trial. They provide leadership, consulting with Sponsors, along with training project teams and investigative sites to ensure our operational strategy is firmly aligned with your scientific and medical objectives. Explore our expert leaders who are shaping ophthalmology research: