High-Quality Central Laboratory Services
Our four wholly owned laboratories offer full-service support to six continents for phase I-IV studies. We have extensive experience from small and simple clinical trials to those trials that are large, global, and complex. Our global laboratory facilities, standardized testing platforms, comprehensive test menu, and stellar project management teams allow Medpace to set up fully customized projects for our clients. Combined with Medpace Clinical Research Organization expertise, we provide a fully integrated solution for your clinical development needs.
Central Laboratory Highlights
Medpace provides laboratory services across 114+ countries delivering best in class science and service supporting today’s complex clinical trials.
- Serving some of the world’s largest, most complex clinical trials
- Extensive capacity and global scalability in state-of-the-art facilities
- Fully harmonized, high throughput instrumentation delivering high quality data
- On-time data transfers with multiple vendor source flexibility to meet client needs
- Biomarker development, validation, and PD analysis
- Industry-leading quick validation timelines
- Web-based data portal – ClinTrak Lab – provides seamless data management delivered in a single integrated global database
Lab Talk: A Video Series with Scientists from Medpace’s Central Laboratories
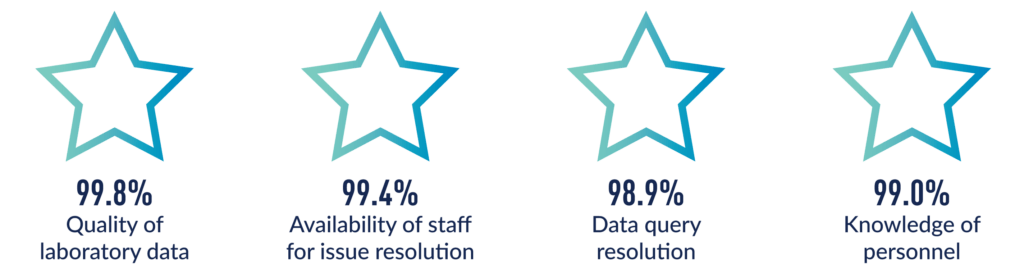
Client Feedback Tells the Story
Your project’s success rests on the quality we deliver. To ensure we are meeting our client’s high-quality expectations, Medpace’s central laboratories compile site satisfaction surveys on a regular basis. This site survey consists of 15 attributes relating to Medpace project performance. The results include feedback from thousands of sites. Medpace earns an average of 99% positive responses (satisfactory or excellent ratings) across all attributes rated.