Our Mission
Accelerating the global development of safe and effective medical therapeutics.
Our unique global partnering philosophy emphasizes an uncompromising commitment to clinical research and to the highest level of ethical standards and performance in our jobs. We are selective about the projects we engage in because we are devoted to quality and providing our partners with best-in-class service.
Medpace’s dedicated teams serve as an extension of your team – we engage quickly and provide strategic thinking – ensuring start-up times are met, superior quality, and the most efficient delivery of every phase of your clinical trial. Our therapeutic and regulatory experts are committed to streamlining your path to approval so every partnership is designed to create research solutions focused on your critical needs.
Why Sponsors Choose Medpace
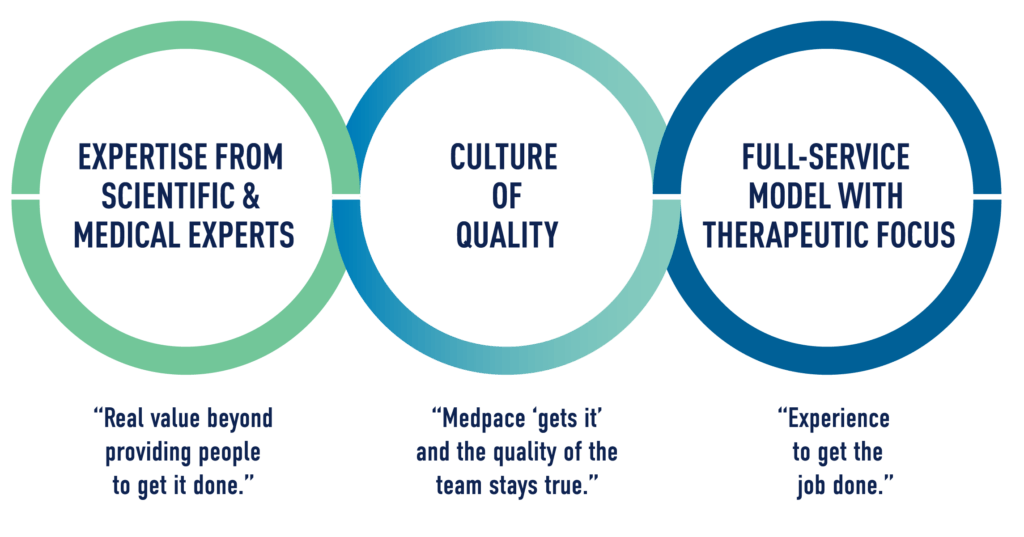
We asked our Sponsors why they chose Medpace as their clinical development partner. Here is what they said:
- Our therapeutically-aligned scientific expertise provides deep insights and leadership throughout the project
- Our culture of quality permeates everything we do
- Our full-service model and solution-oriented project teams deliver consistent and high-quality trial execution