Making the Complex Seamless® in Stroke Clinical Development
Stroke clinical trials are particularly challenging when it comes to patient recruitment and time constraints. Accelerate your stroke study with a clinical research organization (CRO) that specializes in the design and conduct of global trials in neuroscience clinical research, including stroke. As a therapeutically-focused CRO highly experienced in stroke research, Medpace has the medical, regulatory, and operational leadership and disciplined operating approach to position your stroke trial for long-term success.
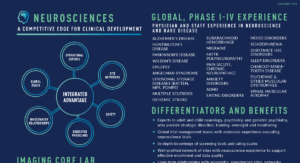
The Medpace Advantage
Gain a competitive edge for your stroke trial by partnering with a global CRO with a proven full-service model that has provided quality results to Sponsors for 30+ years. We have designed and conducted numerous stroke detection and stroke prevention studies. Additionally, our experience includes direct administration of drug to brain, either intraventricularly or intrathecally.
Stroke CRO Capabilities
Partnering with a CRO with a cross-functional collaborative model will position your stroke trial for success. The Medpace Neuroscience Team supports the development of therapeutics for the acute treatment of stroke through the use of our in-house resources including:
- Integrated medical device and diagnostics division with specialized operational and regulatory expertise
- Global central lab with safety and biomarker analysis to support stroke studies
- Industry-leading expertise in cellular and gene therapies including stem cells for acute ischemic stroke
- Deep expertise and experience in vascular research and development, including antithrombotic and lipid lowering therapies
Learn more about our Integrated Approach to Global Clinical Research
Specialized Imaging for Stroke Trials
High-quality image acquisition and interpretation is crucial for the success of trials relying on neuroimaging for patient selection or as a primary endpoint. Medpace Imaging Core Labs, led by radiologists and neurologists with decades of research and clinical experience, supports stroke studies, ensuring imaging components are seamlessly integrated into the complex structure of the overall trial.
Medpace Imaging Core Lab provides an end-to-end suite of imaging services to enhance and expedite biopharmaceutical and imaging contrast agent development. Services include consultation on imaging biomarker strategies, consultation on image acquisition protocols, image collection/archiving services, and image analysis.
Stroke Recruitment and Retention
Successfully recruiting patients for stroke studies requires a partner who has strong relationships with a well-profiled global network of experienced stroke sites, and who can manage complex logistics, negotiations, education, and site maintenance challenges. Medpace has earned a reputation for managing highly challenging studies and has the relationships to work with complex and evolving referral networks (hub and spoke), community hospitals, comprehensive stroke centers, and other acute care settings to drive successful patient enrollment around the world.
Stroke Clinical Trial Experts
Sponsors get the advantage of early and ongoing insight and guidance throughout trial design and execution from a team of scientifically-driven therapeutic experts. Our highly-experienced teams include in-house neurologists, imaging specialists, and operational teams with relevant and recent stroke experience as well as backgrounds in stroke-specific scientific research. Our experience-informed understanding of relevant issues from the perspective of Sponsors, clinical investigators, scientific leaders, and reviewers at regulatory agencies allows us to successfully define and execute clear development plans from beginning to end.
Key Stroke Experience
Medpace has designed and conducted numerous stroke detection and stroke prevention studies. Our biopharmaceutical and medical device experience spans:
- Cellular and gene therapy including stem cell
- Restorative therapies
- Thrombolytic therapies
- Preventative therapies
- Medical device therapies