New White Paper: The Intersection of Rare Diseases and Advanced Therapies and What it Means for Clinical Development
Read the press release for the latest whitepaper on advanced therapies and rare diseases published by Xtalks and Medpace.
Working at Medpace During COVID-19
While COVID-19 has had many effects on how we run our clinical trials as we continue to provide essential services during the pandemic, we’ve also…
What You Missed: The International Liver Conference
The International Liver Congress (ILC) took place digitally this year, but it was worth logging in to keep up-to-date on the novel findings in imaging…
Fall 2020 Virtual Career Fairs
Medpace is excited to partake in another Career Fair season, participating in 20+ virtual events with universities in Ohio, Texas, Indiana and Colorado.
Spotlight on Rare Disease Clinical Development
There is a lot of uncertainty in the world right now, but one thing for certain is that rare disease and orphan indication clinical research…
The First Controlled Clinical Trial For A CRISPR Enhanced Bacteriophage Therapy
In a recent article published in Clinical Leader, Ed Miseta, Chief Editor highlights Medpace's partnership with Locus Biosciences and their development of crPhage™, a bacteriophage…
Webinar: How A Deeper Understanding of Chronic Kidney Disease Is Impacting Clinical Development
Medpace’s experts will be discussing important topics from both a medical and operational perspective.
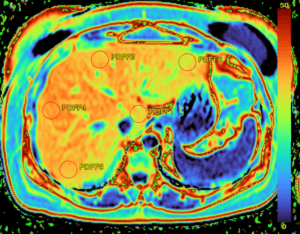
The Evolving Landscape of Therapeutic and Diagnostic Radiopharmaceuticals
In a new article, experts from Medpace’s radiation oncology, imaging, regulatory, and operational teams discuss the growing space of radiopharmaceutical development.
Rare Disease Spotlight: Acromegaly
Learn more about acromegaly and our capabilities for clinical trials.
Operationalizing Complex Innovative Trial Design (FDA’s New Approach)
One of the FDA’s recent initiatives centers on operationalizing complex innovative trial design. Learn more about the types of trials the FDA consider innovative and the…